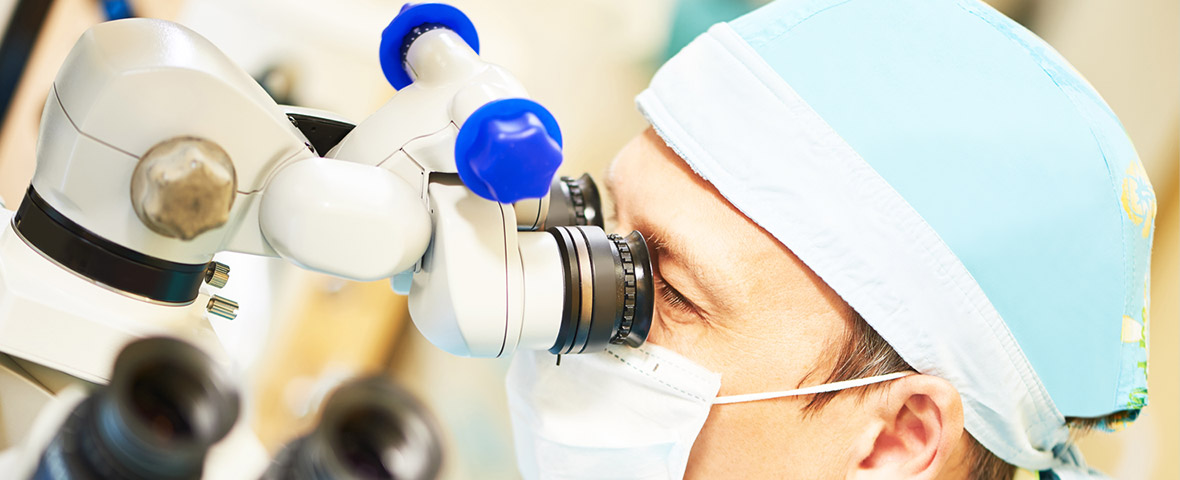
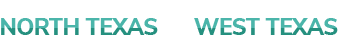Vitrectomy
Some Eye Conditions Require Repair Or Removal Of Material From Within The Eye; E.G., Hemorrhages, Macular Membranes, Floaters, Etc. Very Small Instruments Are Temporarily Placed In The Eye. These Instruments Allow The Physician To Perform The Necessary Tasks To Allow Your Vision To Improve. This Procedure Is Generally Performed In An Outpatient Hospital Setting. After A Short Recovery Time, Patients Are Able To Go Home And Are Commonly Followed Up In The Office The Next Day.
Schedule Your Appointment
How does Avastin Work?
Avastin works by blocking the production of a chemical called “vascular endothelial growth factor” (VEGF). Wet age-related macular degeneration is caused by the growth of abnormal blood vessels in the eye, so by blocking this chemical Avastin can slow the growth of blood vessels that can leak and cause vision loss.
How does Treatment with Avastin Work?
Avastin is administered as an outpatient procedure. Your eye will be cleaned and an anesthetic is applied in order to numb the eye, and the drug is administered using a very fine needle. It is common to need repetitive treatment over time when using Avastin, as repeat treatments are typically necessary for continued results.
EYLEA® (aflibercept) Injection
Indicated for the treatment of patients with neovascular (Wet) Age-related Macular Degeneration (AMD).
Indicated for the treatment of patients with Macular Edema following Central Retinal Vein Occlusion (CRVO).
EYLEA is contraindicated in patients with ocular or periocular infections, active intraocular inflammation, or known hypersensitivity to aflibercept or to any of the excipients in EYLEA.
The Food and Drug Administration (FDA) approved LUCENTIS for the treatment of patients with neovascular or wet macular degeneration. LUCENTIS was specifically developed for intraocular use. It has been developed to treat the underlying cause of wet macular degeneration by targeting the molecular pathway that controls the formation of new blood vessels. LUCENTIS is designed to bind and inhibit the vascular endothelial growth factor (VEGF), a protein that is believed to play a critical role in the formation of new blood vessels. VEGF has been shown to lead to wet macular degeneration progression and central vision loss.
LUCENTIS is a series of injections given directly into the eye every four weeks. Your physician will tell you how long you will need to receive the injections.
To learn more about Lucentis, please click on the link below: